EIQ expands Mayo Clinic agreement ahead of potential FDA clearance for heart failure AI
Our AI heart disease detection Investment Echo IQ (ASX: EIQ) just expanded its agreement with the Mayo Clinic, one of the top-ranked hospital systems in the US.
EIQ and Mayo clinic expanded an agreement that would see EIQ’s Ai Heart Failure detection sold by Mayo through its network of hospitals IF/When the FDA provides clearance on the tech.
Basically, EIQ has signed a reselling/distribution deal BEFORE FDA clearance…
For one of the top-ranked Hospital networks in the US (across 80+ external partner hospitals).
The reason we think today’s news matters is because the agreement is with the same clinic that ran the validation studies for EIQ.
The validation studies showed EIQ’s Heart Failure tech demonstrated a sensitivity of 99.5% in identifying patients with heart failure and a specificity of 91.1% in correctly identifying patients without heart failure.
We covered those in November in this article: EIQ: Announces its AI tech can detect Heart Failure too - with 99.5% sensitivity and 91.0% specificity
Clearly, Mayo Clinic is impressed by those results - enough so that they are signing a distribution deal even before FDA clearance.
We think that third party validation of the strength of the results (and fingers crossed is a good omen for FDA clearance).
(important to note that these results have not been reviewed or cleared by the FDA and there is no guarantee of clearance, clearance is due as soon as this month)
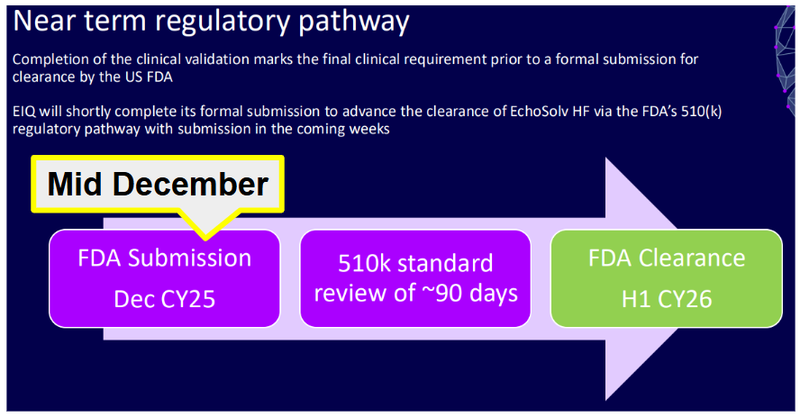
EIQ has a 510(k) application under review with the FDA right now and expects to have clearance as soon as this month:
(Source)
The FDA submission was made in mid December and with the standard review time of around 90 days (from the slide above), it could be due any day now, although EIQ guide during H1.
For context EIQ already has FDA clearance for its first product which detects aortic stenosis (a heart valve disease). (source)
Aortic Stenosis is a heart disease which costs the US healthcare system ~US$10BN per annum, so it's a big market. (source)
The big catalyst for EIQ is coming inside the next few months (or even weeks)…
In December last year, EIQ made its submission to the FDA, which we covered here.
We think FDA approvals for heart failure could be a major catalyst for EIQ because:
- Only 50% of heart failure cases are accurately diagnosed
- 1 in 4 Americans are impacted by heart failure over their lifetime.
- Heart failure is the leading cause of rehospitalisation and accounts for 17% of US healthcare expenditure. AND
- The heart failure treatment market is worth ~US$60BN…
A mentioned above, late last year EIQ released the clinical validation study results for its heart failure tech which showed:
- Sensitivity of 99.5% (The ability to identify 99.5% of all patients who actually had heart failure). (source)
- Specificity of 91.0% (the ability to identify 91% of patients who don't have heart failure). (source)
Basically this means that when EIQ’s tech is plugged into cardiologists' workflows, IF the AI says you are positive... 99.5% of the time, the AI is correct...
Which is much better than the only other Ai heart failure detection software used in the market today, which delivers - sensitivity of 87.8% and specificity of 81.9% (source).
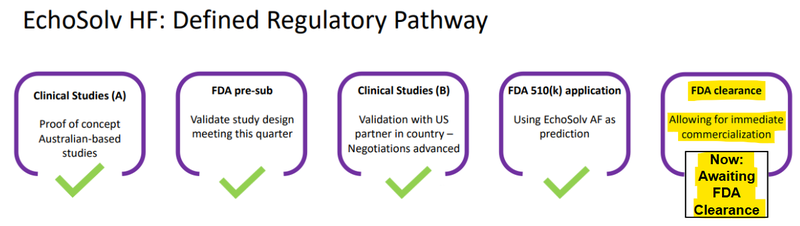
So now with the expanded agreement, should EIQ get FDA approval, it now has a clear pathway already defined to get it in use.
(Source)
EIQ’s guidance was for a decision in H1-2026 so we could see some news on the FDA clearance any day now…
Another reason we think the FDA clearance is a major catalyst for EIQ is because of the existing reimbursement pathway (pathway to revenue generation for EIQ) that EIQ can piggyback off of. (we covered that in our last note here).
IF EIQ gets FDA clearance it can go on commercialising the tech almost immediately (especially with agreements like the one signed today).
What’s next for EIQ?
🔄 Heart failure FDA approvals
Next is all about FDA clearance for EIQ’s heart failure detection tech.
As mentioned above, we are expecting to hear back from the FDA inside H1-2026… possibly as early as this month
🔄 Commercialisation updates for Aortic Stenosis AI tech
The key metric we will be tracking in the short term is how many integrations EIQ can secure for its Aortic Stenosis tech.
In the short term we want to see more distribution deals - either through strategic partnerships or reseller deals.
Today we got an update related to this in the form of an expanded agreement with the Mayo Clinic to include its heart failure detection software, so should FDA clearance come through on this, it can start to be commercialised immeadiately.
🔄 Australia and NZ pilot program
EIQ has previously mentioned that this program is being run with a ”leading global structural heart innovation company”.
We want to see some more news on this front because we think it could help advance EIQ’s licensing revenue pathway and be a “proof of concept” study that EIQ can take into the US.
🔄 Partnership with European reseller to broaden market exposure
We want to see EIQ expand into new markets like Europe, in a previous webinar EIQ said the company was pursuing this opportunity and last week the company updated that there was progress on this with negotiations underway.
This also includes CE Mark and TGA applications so that EIQ can sell into Europe and Australia.






