ILA signs an agreement with a US Army adjacent research institute.
Our 2025 biotech Pick Of The Year Island Pharmaceuticals (ASX: ILA) just signed a 3 year Cooperative Research and Development Agreement with:
- USAMRIID, the US Army’s premier infectious disease research institute who operate Biosafety Level 4 facilities - capable of working with viruses like Marburg disease.
- The Geneva Foundation, a highly influential non-profit that manages nearly US$383M in military medical research funding and helps navigate Department of War (DoW) requirements.
… To accelerate the “final stages” of the animal rule FDA approvals process for its Marburg disease drug.
Basically, today’s partnership would give ILA access to the infrastructure (Level 4 labs) and expertise needed to execute the final stages of development of its drug.
ILA recently received approval from the FDA to utilise the “Animal Rule” for its Marburg drug (Galidesivir).
The Animal Rule drug approval pathway cuts years off the usual FDA approvals process because of how deadly these conditions can be (and the urgency around defending against bioterrorism and bioweapon threats).
For context - Marburg is classified as a Category A bioterrorism threat (the highest level threat) by the US government (source).
And Marburg is the only Category A biothreat with NO current vaccine or FDA approved treatment...
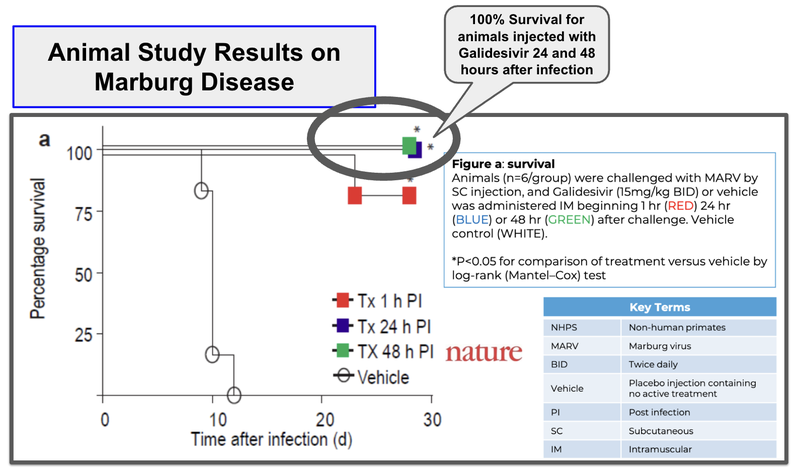
ILA’s drug already has human safety trials completed.
And has existing animal trials showing 100% survival rates for animals treated at 24-48 hours after infection, compared to the placebo which showed 0% survival.
(Source)
ILA is now in the final two stages of studies which requires:
- An optimisation study (firstly to to work out the optimal dose and when the best time to administer that dose is)
- A pivotal confirmatory study (when we see how effective ILA’s Marburg drug really is)
IF successful, ILA could unlock:
- A Priority Review Voucher (PRV) that comes in if ILA’s Galidesivir is FDA approved - a recent PRV sale by Jazz Pharma was for US$200M. (source)
- US Government national stockpiling deals - these can be worth anything between US$100M to US$1.2BN ANNUALLY (source)
The fast tracked approval pathway, PRV potential and the potential stockpiling deals is a big part of the reason why we made ILA our 2025 biotech pick of the year.
We think today’s announcement is the equivalent of early engagement with the same organisations that could help ILA become a stockpiling contract candidate.
Adding to ILA’s existing partnerships - ILA recently hired US advisors (Todd Strategy Group) and joined the Medical Countermeasures Coalition (MC2) to advance US government engagement initiatives.
Some well established companies are a part of the MC2 group including SIGA Technologies and Bavarian Nordic - both of which have received “Strategic National Stockpile” contracts:

(source)
What’s next for ILA in the next 6-9 months
Now that ILA has been given the green light to pursue approvals via the Animal Rule AND the FDA has confirmed a staged approach would be appropriate we want to see ILA execute the following:
- 17th November: ILA confirms Animal Rule eligibility for its Marburg drug ✅ (source)
- Recently: staged approach for FDA approvals confirmed ✅ (source)
- NEXT: We want to see ILA start “optimisation studies” ahead of a pivotal study later this year. Today’s update relates to helping set up and begin this
- NEXT: We want to see ILA sign more agreements with Biosecurity Level 4 (BSL4) facilities that are able to run animal studies - more sites means the studies can be completed quicker… Today’s update relates to helping set up and begin this step also
- THEN, we want to see ILA start animal trials (pivotal trial) for deadly Marburg disease. (this is the big one)
Here are the milestones we will be tracking for the animal study:
- 🔲 Clinical trial design completed
- 🔲 Clinical trial starts
- 🔲 Clinical trial completed
- 🔲 Clinical trial results
Assuming the clinical trial results are positive, ILA will then submit to the FDA for an Investigational New Drug (IND) approval of its drug (6 month review timeframe)
IF APPROVED…
ILA COULD SECURE a Priority Review Voucher, which is a tradeable asset worth on average ~US$100M (think of this like a ‘thank you’ for developing the drug)
ILA COULD SECURE commercial stockpiling contracts with the US Department of Defence for protection against bioweapons. These contracts can be very lucrative.
In a recent webinar, Chairman Jason Carroll also gave a pretty good run down of what’s to come for ILA.

Watch it here - ILA - COMBATTING URGENT VIRAL DISEASE THREATS