IIQ pairs up with prestigious Peter Mac on potential breakthrough breast cancer research (with a string of catalysts to follow)
Disclosure: S3 Consortium Pty Ltd (the Company) and Associated Entities own 1,150,000 IIQ Shares and 575,000 IIQ Options at the time of publishing this article. The Company has been engaged by IIQ to share our commentary on the progress of our Investment in IIQ over time.
Yesterday, our biotech Investment Inoviq (ASX:IIQ) signed one of its most important research deals to date.
IIQ is a cancer diagnostics and therapeutics company developing an exosome platform technology.
Exosomes are tiny, microscopic extra-cellular “containers” released by ALL human cells.
Exosomes can carry ‘messages’ - clues as to whether or not someone has a disease - and increasingly it looks like they can also be successfully ‘weaponised’ - which is how IIQ is able to locate and kill cancer cells in the lab.
IIQ will soon be working with the Peter MacCallum Cancer Centre (Peter Mac) in Melbourne to help validate its CAR-exosome therapy for triple negative breast cancer (TNBC).
(and potentially other cancer types in the future...)
With over 47,000 patients, Peter Mac brings more than research prestige, it provides IIQ with access to advanced infrastructure, well practiced preclinical models, and decades of experience in translational cancer research.
The institution is among only a handful globally that combines a dedicated cancer hospital with a fully integrated cancer research program.
That means there is a ready made cohort of patients to which cutting edge treatments can be quickly deployed in a clinical setting.
IIQ’s therapy has already been shown to kill 75% of breast cancer cells in a test tube within just 72 hours.
IIQ demonstrated the effectiveness of the technology only in June of last year.
Interest in the technology helped take the IIQ share price from ~46.5c to a peak of 79.5c in the space of a month - today the IIQ share price sits at ~40.5c.
This potential “moonshot” cancer therapy was one of the key reasons we initially Invested in IIQ, just after those results came out.
Fast forward to today, and Peter Mac will now be working with IIQ to advance this therapy from its current preclinical stage - to ideally, a state of readiness for a clinical trial with actual cancer patients.
By partnering with Peter Mac, we see this as important validation for IIQ’s preclinical work on this cancer therapy.
Peter Mac doesn’t work with just any company, so they must see promise in what IIQ is doing.
As far as we can tell, there are only a handful of small caps and pre-IPO companies that are working with Peter Mac (outside of the huge Big Pharma names like Bristol-Myers Squibb, GlaxoSmithKline and Roche).
Peter Mac is a prestigious research organisation and we hope that IIQ’s cancer therapy can now progress through additional in vitro (test tube) and in vivo (animal) studies.
Results from the lab studies (in vitro) with Peter Mac are expected in Q3 2025.
Results from the mice studies (in vivo) are expected in Q4 2025.
Then the path to clinical trials (humans) opens up.
And that’s where the big re-rate could happen...
(not to mention all the other big catalysts IIQ has planned for this year - more on those below).
All going well, IIQ can then begin preparations for clinical trials (with humans) - and we’ve seen pre-clinical work like this leading into clinical trials deliver major share price re-rates before too.
One of our other Investments, Arovella Therapeutics went from ~2.5c to a high of ~20.5c on preclinical research success.
That is a ~720% move without ever going into human studies...
Of course, the past performance of one company should not be taken as an indication of future performance in another company.
The Peter Mac agreement is just the “tip of the iceberg” in terms of newsflow to come from IIQ too...
Today for instance, we saw IIQ release peer-reviewed evidence that its breast cancer monitoring test is remarkably more accurate than the leading FDA approved test (a test by $483BN Roche).
...by more than 25%.
(keep reading for more on that below, breast cancer monitoring is a US$668M market)
Clearly, there’s a lot going on for IIQ - here’s quick summary of the brimming list of upcoming potential catalysts for the company:
- 🔄Further updates on solid tumour therapy in vivo studies (targeting Q3 in vitro results and Q4 in vivo results with Peter Mac this year)
- 🔄A lab partner for SubB2M tests (for breast cancer monitoring - expected this quarter)
- 🔄Clinical validation studies for highly accurate ovarian cancer screening test (expected this quarter)
- 🔄 Development of exosome diagnostic for neurodegenerative conditions like Alzheimer’s and Parkinson's. (diagnostic development to start in Q3 of this year)
IIQ will be doing an investor briefing to cover the company’s latest developments on Thursday this week, 3rd April 2pm AEDT:
Click here to register for the IIQ Investor Briefing
We’ll hear from IIQ CEO Dr Leearne Hinch, Advisory Board member Prof Phil Darcy (who works at Peter Mac by the way) and Dr Emma Ball, IIQ new Chief Commercialisation Officer (more on her background below).
While we await that briefing, in this note we will cover:
- Some examples of how Peter Mac has long been at the forefront of new cancer treatments
- What the Peter Mac and IIQ partnership is all about
- How much bigger the cancer therapeutics market is relative to diagnostics (almost 10x bigger) and how IIQ fits in
- How does IIQ’s breast cancer killing CAR-exosome therapy work?
- Our Big Bet for IIQ
- What catalysts lie ahead for IIQ
- IIQ’s new Chief Commercial Officer, Dr Emma Ball and
- IIQ’s new medical and scientific advisory board
Let’s dive in...
Peter Mac has long been at the forefront of new cancer treatments
As a globally recognised cancer fighting institution, Peter Mac has 75 years of history.
Peter Mac has brought new technology to bear on the disease, while also conducting groundbreaking clinical trials that have reshaped cancer treatment.
Here is just a small sample of some of the milestones in its rich history:
- In 1956 Peter Mac introduced a linear accelerator to Australia for cancer treatment (at the time, a new fangled radiation therapy device for deep tumours) (Source)
- 2010, saw the advent of robotic assisted surgery in Australia at Peter Mac (Source)
- Last year, Peter Mac led a Phase 3 international clinical trial where, five years after treatment, 60% of a specific form of lung cancer patients that were treated with a drug survived - a remarkable jump in the usual disease progression. (Source)
What will Peter Mac and IIQ be working on?
The first specific cancer Peter Mac and IIQ will be working together on fighting is Triple Negative Breast Cancer (TNBC).
TNBC is a subtype of breast cancer - and one of the worst cancers out there.
It lacks three main receptors used in most breast cancer diagnostics and treatments, estrogen, progesterone, and the protein HER2.
Receptors are little markers on cancer cells, and receptors like these help clinicians diagnose breast cancer.
If you can’t find the cancer with these receptors, it often gets caught too late.
Triple Negative Breast Cancer accounts for ~15% of all breast cancer cases but has disproportionately negative outcomes due to limited treatment options and high recurrence rates.
Triple Negative Breast Cancer is also a solid tumour - solid tumours make up ~90% of all cancers.
Unlike other breast cancer types, Triple Negative Breast Cancer does not respond to hormone or HER2-targeted therapies, leaving chemotherapy as the primary option.
In our last IIQ note, the company’s therapy was able to kill 30% of triple negative breast cancer cells (a particularly aggressive type of cancer) in a test tube:
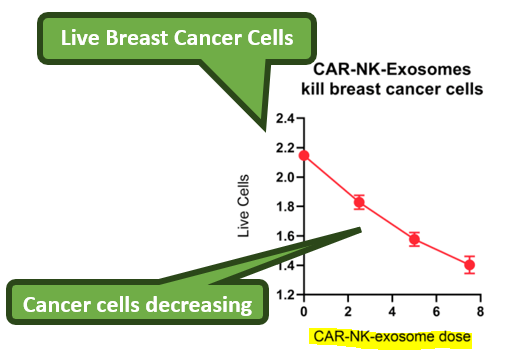
This is from just a “Proof of Concept” in vitro (test tube) study which IIQ released data from in December of last year.
More lab data is expected in Q3 2025.
Then mice (that is due in Q4 of this year)
Then the path to clinical trials (humans) opens up.
And that’s where the big re-rate could happen (not to mention all the other big catalysts IIQ has planned for this year - more on that in a moment).
The cancer therapeutics market is almost 10x bigger than diagnostics... here’s how IIQ fits in
IIQ is developing a platform technology which uses exosomes.
IIQ was initially focussed on diagnostics, but increasingly IIQ is aiming to “weaponise” exosomes in a new approach to cancer therapeutics.
Before we explain why, let's establish a few definitions.
Short Biotech 101 Lesson - Diagnostics vs Therapeutics:
If the main objective is to establish a diagnosis for a patient prior to starting any kind of treatment, then it is considered to be a diagnostic.
If a diagnosis is already established and a procedure is required in order to eradicate a problem, then it is considered to be a therapeutic.
Cancer therapeutics (i.e. getting rid of cancer) is a much larger market than diagnostics (merely confirming or otherwise if someone has cancer), so this business strategy makes sense too.
And while it is extremely important to diagnose cancer early - treating cancer is where the majority of capital inflows will happen for early stage biotechs in the space...
This is reflected in the following market values for the areas IIQ operates in:
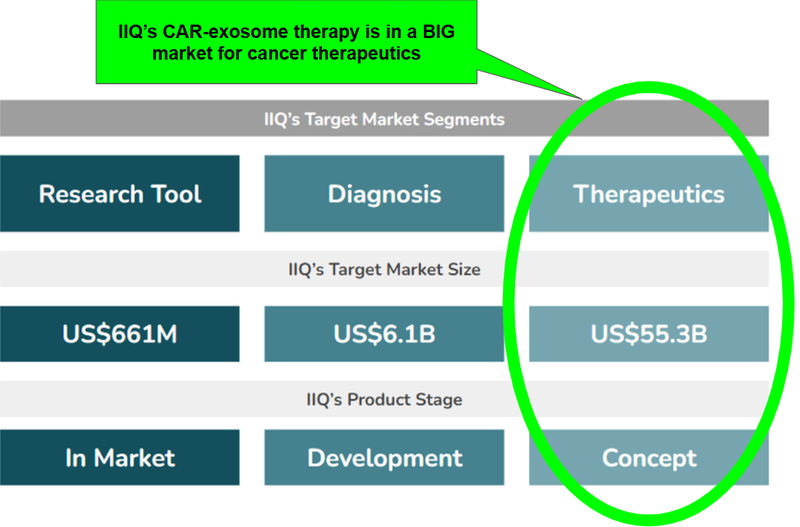
OK now the market sizes are covered, here’s our simple explanation of how IIQ’s therapy works, using a couple analogies which we find helpful for understanding the technology.
How does IIQ’s breast cancer killing CAR-exosome therapy work?
As many of us are unfortunate to know, chemotherapy is like burning down a house to kill a cockroach.
It’s incredibly painful for patients and their loved ones.
IIQ’s therapy offers a novel approach to the problem: it uses exosomes.
Think of exosomes as the postal service in the human body - they deliver packages with precise instructions and information, helping cells communicate.
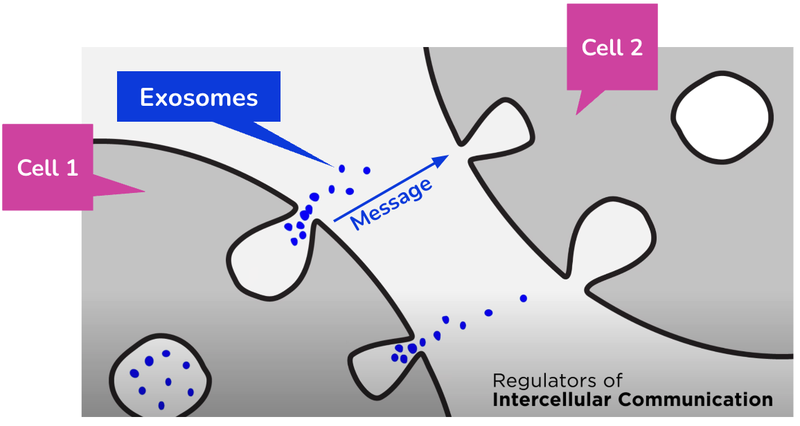
While exosomes are primarily used for cell to cell communication, they can also act as a sort of cellular special forces...
Finding out where the bad guys are (cancer and other diseases) and what they are up to.
But because they travel around so much and can move between cells - it is believed that they can also deliver powerful therapies that can kill bad guys (cancer and other diseases) as well.
A good guy that is small enough to find the bad guy's - and can also bring the bad guy to justice.
Control the “messenger,” find out what’s happening in the cell AND (hopefully) get the messenger to deliver potentially life saving therapies.
That’s what IIQ is showing its technology is capable of doing.
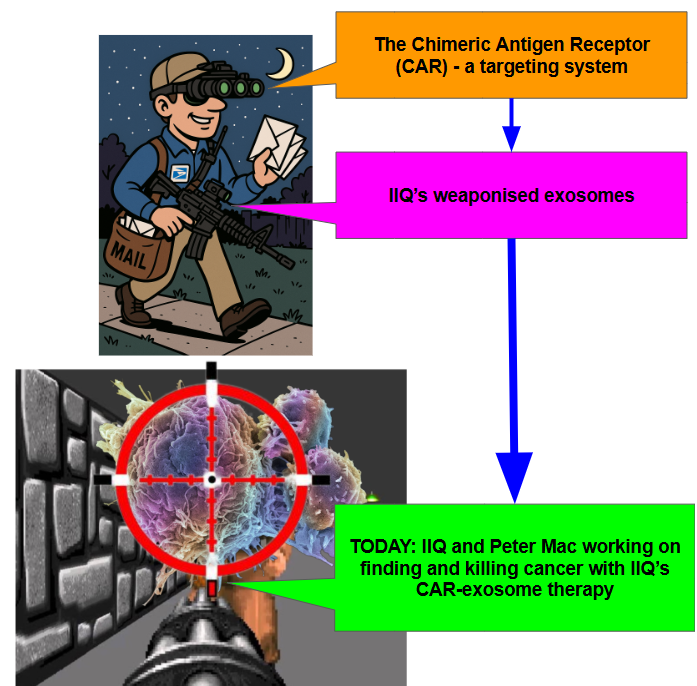
IIQ is combining its exosome technology with Chimeric Antigen Receptor or CAR.
CARs have been used in a number of different cell therapy platforms - it is a specific receptor that allows the molecule to identify the cancer cell.
Think of a CAR as a targeting device that helps the body's immune system find and kill cancer cells.
Normally, cancer hides from the immune system, but CARs give cells the ability to recognise specific cancer markers.
Once they see the target, they zero in and destroy it.
Combining exosomes with the CAR is like the postman is now armed, and knows who the bad guys are.
Here’s our super crude representation of what IIQ’s CAR-exosome therapy is doing with its preclinical work in conjunction with Peter Mac:
With this technology, IIQ can now look forward to data releases in the coming two quarters - and solid tumours make up just one option for IIQ’s multiple “shots on goal” that we think can provide a large sustained re-rate.
We’ve captured these “shots on goal” in our ultimate upside scenario for our IIQ Investment:
Our Big Bet for IIQ
“IIQ re-rates to a +$500M market cap on commercialisation of its breast cancer test, its ovarian cancer test, neurodegenerative disease test and/or its solid tumour (breast cancer) therapy.”
NOTE: our “Big Bet” is what we HOPE the ultimate success scenario looks like for this particular Investment over the long term (3+ years). There is a lot of work to be done, many risks involved - just some of which we list in our IIQ Investment Memo. Success will require a significant amount of luck. There is no guarantee that our Big Bet will ever come true.
IIQ has plenty to come this year...and multiple catalysts
IIQ has a lot of strings to its bow.
In particular we are looking forward to the following potential catalysts:
- 🔄 Yesterday:Further updates on solid tumour therapy in vivo studies (targeting Q3 in vitro results and Q4 in vivo results with Peter Mac this year)
- 🔄 A lab partner for SubB2M tests (for breast cancer monitoring - expected this quarter)
On the SubB2M front - IIQ has a really accurate breast cancer monitoring test.
Last year a clinical validation study of IIQ’s breast cancer test showed that it detected 19% more breast cancers than the leading test (CA15-3 test by $483BN Roche) with an impressive 81% sensitivity and 93% specificity across a range of breast cancers.
Importantly, this data has been corroborated by today’s IIQ announcement which details the publication of a scientific paper in the journal Breast Cancer Research and Treatment:

(Source)
Having peer-reviewed evidence that your breast cancer monitoring test is more accurate than the leading FDA approved test (Roche’s test) can only be a good thing in terms of future commercialisation efforts.
What’s more - the sensitivity and specificity of the stage test I and stage II breast cancers “compares favourably to mammography” - these are the breast exams administered by other clinicians.
That is quite remarkable - and could make potential lab partners sit up and take notice of IIQ’s blood test as a viable alternative to not just the FDA approved Roche test, but potentially the time consuming task of mammography.
Just on that lab partner we mentioned...
A lab partner would help take IIQ closer to commercialising this test as the partner would begin to use the test in “real-world” clinics - where tests are performed everyday.
These are busy labs as breast cancer monitoring has to be done usually four times a year.
Given the common occurrence of breast cancer and the need to stay on top of it, commercialisation for IIQ in this potential market here is worth around US$668M (Source - slide 17)
Recent deal values over the last few years for companies developing blood tests that accurately identify cancer range from US$250M in upfront payments to upwards of US$450M.
All multiples of IIQ’s current market cap.
Read: News in the queue for IIQ - upcoming share price catalysts...
- 🔄 Clinical validation studies for highly accurate ovarian cancer screening test (expected this quarter)
IIQ also has a potential breakthrough on its hands in ovarian cancer screening - bear in mind there is no blood test for ovarian cancer screening test available today, and testing usually requires a biopsy and an ultrasound.
So further news on that front could be very promising for IIQ.
Read: IIQ announces extremely accurate cancer screening test - 94%
- 🔄 Development of exosome diagnostic for neurodegenerative conditions like Alzheimer’s and Parkinson's. (diagnostic development to start in Q3 of this year)
Finally, IIQ has been working on a very important task for patients and the medical community more broadly - a way to detect Parkinson’s and Alzheimer’s early.
Both neurodegenerative diseases are increasingly prominent in society with our aging populations.
Currently, there is no lab or imaging test that is recommended or definitive for Parkinson’s disease. (Source)
IIQ could be able to produce a further scientific breakthrough using NEURO-NET, its product that is being developed to find the “fingerprints” of these two diseases:
How does yesterday’s news impact our IIQ Investment Memo?
Yesterday’s update is directly related to one of the key reasons we Invested in IIQ from our Investment Memo:
Early stage moonshot to treat solid tumours
IIQ is also advancing a solid tumour therapy which has been shown to kill 75% of breast cancer cells in 72hrs in an in vitro (test tube) study. There is a huge unmet need for treatments in the solid tumour space where, so far, cell therapies haven’t been developed.
Source: “Why did we Invest in IIQ?” - IIQ Investment Memo 12 June 2024
We think any preclinical success for IIQ on this “moonshot” treatment for solid tumours - in this case IIQ is targeting particularly nasty Triple Negative Breast Cancer cells - could help IIQ re-rate if it is able to move closer to the clinical stage.
This agreement with Peter Mac should certainly help IIQ in that regard.
IIQ brings on Chief Commercial Officer, Dr Emma Ball
A few weeks ago, IIQ appointed Dr Emma Ball as its new Chief Commercial Officer (CCO).
Dr Ball is an industry veteran with 25 years of experience across prominent roles at major companies like $20BN capped NASDAQ-listed Illumina and $120BN capped Aussie biotech giant CSL.
We see this as a strong appointment as IIQ gets closer to market entry and commercialisation across its portfolio of highly promising cancer diagnostics and therapeutics as well as its exosome research products.
We think this is important as it provides IIQ with strong commercialisation acumen as it pursues potential licensing deals across its portfolio
The March announcement notes:
“[Dr. Ball] led search and evaluation, due diligence and negotiations for multiple transactions including research collaborations, co-development, in- and out-licensing deals, and mergers, acquisitions and divestments.”
We like the sound of that, particularly considering some of the potential in IIQ’s product portfolio - not least of which is a breast cancer monitoring test.
IIQ has also established a medical and scientific advisory board, and there’s a connection
Prior to the appointment of Dr Ball, in February IIQ announced the establishment of its scientific advisory board.
Among the newly appointed experts in IIQ’s are:
- Professor H. Miles Prince AM: Leading Clinical Haematologist and Oncologist and Professor at both Melbourne and Monash universities. He is an NHMRC Investigator Fellow and has been principal investigator of over 100 clinical trials including targeted therapeutics (CAR-T therapy) for haematological conditions and cancers.
- Professor Phillip K. Darcy: Group Leader of the Cancer Immunotherapy Laboratory at the Peter MacCallum Cancer Centre and NHMRC Principal Research Fellow, focusing on novel T cell-based immunotherapy approaches for cancer in preclinical mouse models and clinical translation.
- Professor Carlos Salomon: Director of the University of Queensland Centre for Extracellular Vesicle Nanomedicine, Head of the Translational Extracellular Vesicles in Obstetrics and Gynaecology Group and NHMRC Investigator Fellow, specialising in exosome biology and its clinical translation to diagnostics and therapeutics for ovarian cancer and obstetrical syndromes.
- Dr James McCracken: Leading Medical Oncologist specialising in breast cancer treatment at Epworth Healthcare and the Peter MacCallum Cancer Centre. His research interests include the field of liquid biopsies for cancer to personalise treatment and minimise toxicity.
Of particular note for us is the presence of Professor Phillip K. Darcy who is Group Leader of the Cancer Immunotherapy Laboratory at the Peter MacCallum Cancer Centre.
He’ll be presenting during IIQ’s investor briefing this week (register for that here) and the fact that he is currently working at Peter Mac and will now be working with IIQ adds further weight to IIQ’s preclinical work on a CAR-exosome therapy.
Beyond Professor Darcy, IIQ’s new panel of experts have a strong mix of skills, across cancer therapeutics and diagnostics, exosomes and liquid biopsies.
We think these experts will be particularly valuable to IIQ as it seeks to successfully knock down multiple catalysts in the coming quarters, ideally helping the company achieve a sustained re-rate.
What could go wrong?
While the rewards on success can be extremely large, all small cap biotech investments are risky investments.
The main risk relevant to yesterday’s announcement from IIQ is “technology risk”.
IIQ expects to be taking its therapeutic product “in vivo” (animal studies) in Q4-2025.
There is always a risk that once the therapeutic goes into animal studies, it isn't as effective or doesn't work at all.
If the tech isn't able to replicate the same (or better) results in animal studies then it could re-rate the IIQ share price lower.
Technology risk
Exosomes are still an emerging field of scientific research and there may be undiscovered drawbacks to exosomal products - in particular as they apply to therapeutics.
For example, IIQ’s preclinical research on exosome therapies may not yield the desired results or may not be deemed acceptable in the eyes of a regulatory body.
Source: “What could go wrong?” - IIQ Investment Memo 12 June 2024
To see more risks, check out our IIQ Investment Memo here.
Our IIQ Investment Memo
Our Memo provides a short, high-level summary of our reasons for Investing. We use this memo to track the progress of all our Investments over time.
In our IIQ Investment Memo, you can find the following:
- What does IIQ do?
- The macro theme for IIQ
- Our IIQ Big Bet
- What we want to see IIQ achieve
- Why we are Invested in IIQ
- The key risks to our Investment Thesis
- Our Investment Plan
General Information Only
This material has been prepared by StocksDigital. StocksDigital is an authorised representative (CAR 000433913) of 62 Consulting Pty Limited (ABN 88 664 809 303) (AFSL 548573).
This material is general advice only and is not an offer for the purchase or sale of any financial product or service. The material is not intended to provide you with personal financial or tax advice and does not take into account your personal objectives, financial situation or needs. Although we believe that the material is correct, no warranty of accuracy, reliability or completeness is given, except for liability under statute which cannot be excluded. Please note that past performance may not be indicative of future performance and that no guarantee of performance, the return of capital or a particular rate of return is given by 62C, StocksDigital, any of their related body corporates or any other person. To the maximum extent possible, 62C, StocksDigital, their related body corporates or any other person do not accept any liability for any statement in this material.
Conflicts of Interest Notice
S3 and its associated entities may hold investments in companies featured in its articles, including through being paid in the securities of the companies we provide commentary on. We disclose the securities held in relation to a particular company that we provide commentary on. Refer to our Disclosure Policy for information on our self-imposed trading blackouts, hold conditions and de-risking (sell conditions) which seek to mitigate against any potential conflicts of interest.
Publication Notice and Disclaimer
The information contained in this article is current as at the publication date. At the time of publishing, the information contained in this article is based on sources which are available in the public domain that we consider to be reliable, and our own analysis of those sources. The views of the author may not reflect the views of the AFSL holder. Any decision by you to purchase securities in the companies featured in this article should be done so after you have sought your own independent professional advice regarding this information and made your own inquiries as to the validity of any information in this article.
Any forward-looking statements contained in this article are not guarantees or predictions of future performance, and involve known and unknown risks, uncertainties and other factors, many of which are beyond our control, and which may cause actual results or performance of companies featured to differ materially from those expressed in the statements contained in this article. S3 cannot and does not give any assurance that the results or performance expressed or implied by any forward-looking statements contained in this article will actually occur and readers are cautioned not to put undue reliance on forward-looking statements.
This article may include references to our past investing performance. Past performance is not a reliable indicator of our future investing performance.

